【International Papers】Thermodynamic and Experimental Study of β-Ga₂O₃ Growth by Oxide Vapor-Phase Epitaxy using Ga₂O₃ and H₂O as Precursors
日期:2026-04-13阅读:96
Researchers from the Sophia University have published a dissertation titled " Thermodynamic and Experimental Study of β-Ga2O3 Growth by Oxide Vapor-Phase Epitaxy using Ga2O3 and H2O as Precursors" in Japanese Journal of Applied Physics.
Background
β-Gallia-structured Ga2O3 (β-Ga2O3) has attracted considerable attention as a promising material for future high-power electronic devices, because its Baliga’s figure of merit is significantly larger than that of other wide-bandgap semiconductors such as GaN and 4H-SiC. This superiority originates from its wide bandgap of 4.4-4.9 eV, which yields a large dielectric breakdown field of approximately 8 MV/cm together with a theoretically predicted maximum room-temperature electron mobility of ~200 cm2/Vs. Large diameter single-crystal β-Ga2O3 wafers have also been fabricated by melt-growth techniques such as the floating zone (FZ), edge-defined film-fed growth (EFG), Czochralski and vertical Bridgman (VB) methods.
Abstract
Thermodynamic analysis of β-Ga2O3 growth by oxide vapor-phase epitaxy (OVPE) usingGa2O and H2O precursors was conducted, supported by preliminary growth experiments. Calculations clarify practical growth windows as functions of temperature, the H2 mole fraction in the carrier gas, the input VI/III ratio, and the input partial pressure of Ga2O (POGa2O).The analysis revealed that sustained β-Ga2O3 growth above 1000 °C requires careful control of the H2 mole fraction to balance precursor generation and thermodynamically driven etching. Phase diagrams were constructed as functions of temperature, POGa2O, and input VI/III ratio, which delineate the growth and etching regimes. β-Ga2O3 was grown on (0001)sapphire substrates, and the experimentally derived driving forces agree well with thethermodynamic predictions. The results demonstrate that β-Ga2O3 growth using Ga2O and H2O can be effectively controlled based on thermodynamics, offering a chloride-free, safer alternative for power-device-grade drift layers.
Highlights
A novel chlorine-free OVPE growth system was developed, which for the first time enabled the OVPE growth of β-Ga₂O₃ using Ga₂O and H₂O as dual precursors.
A complete thermodynamic theoretical system for this chlorine-free system was established, revealing the coupled effect of H₂ on precursor generation in the source zone and thermodynamic equilibrium in the growth zone.
A precise match between thermodynamic theory and experiments was achieved, verifying the controllability of the system and making up for the deficiencies of traditional methods in high-temperature growth regulation.
Conclusion
This novel oxide vapor phase epitaxy method offers significant advantages including improved safety, elimination of reactor corrosion, and absence of CI incorporation issues. The good agreement between thermodynamic calculations and experimental results demonstrates that β-Ga2O3 growth using Ga2O and H2O can be effectively controlled based on thermodynamic principles. Future studies will focus on detailed material characterization and device fabrication to fully realize the potential of this growth technology for β-Ga2O3 power device applications.
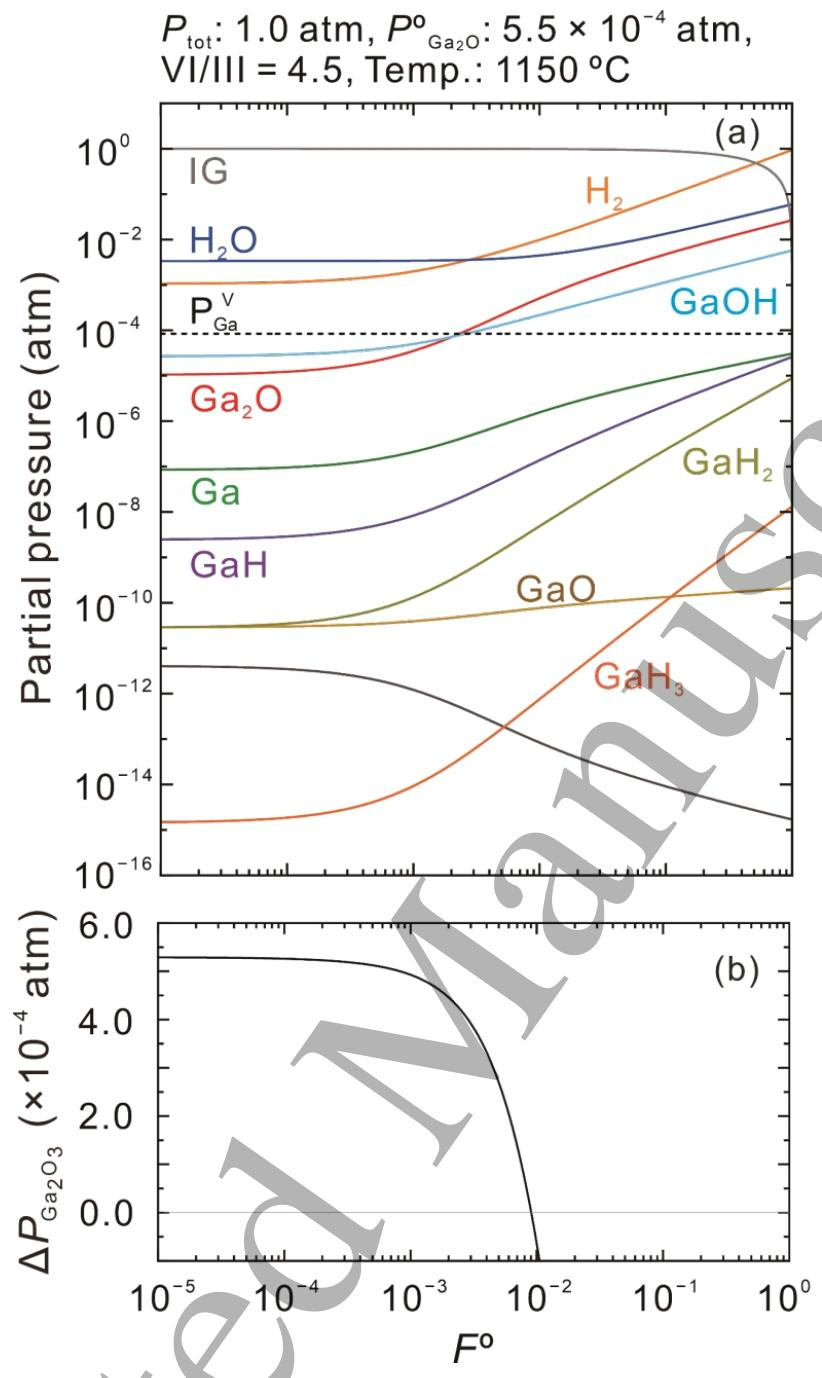
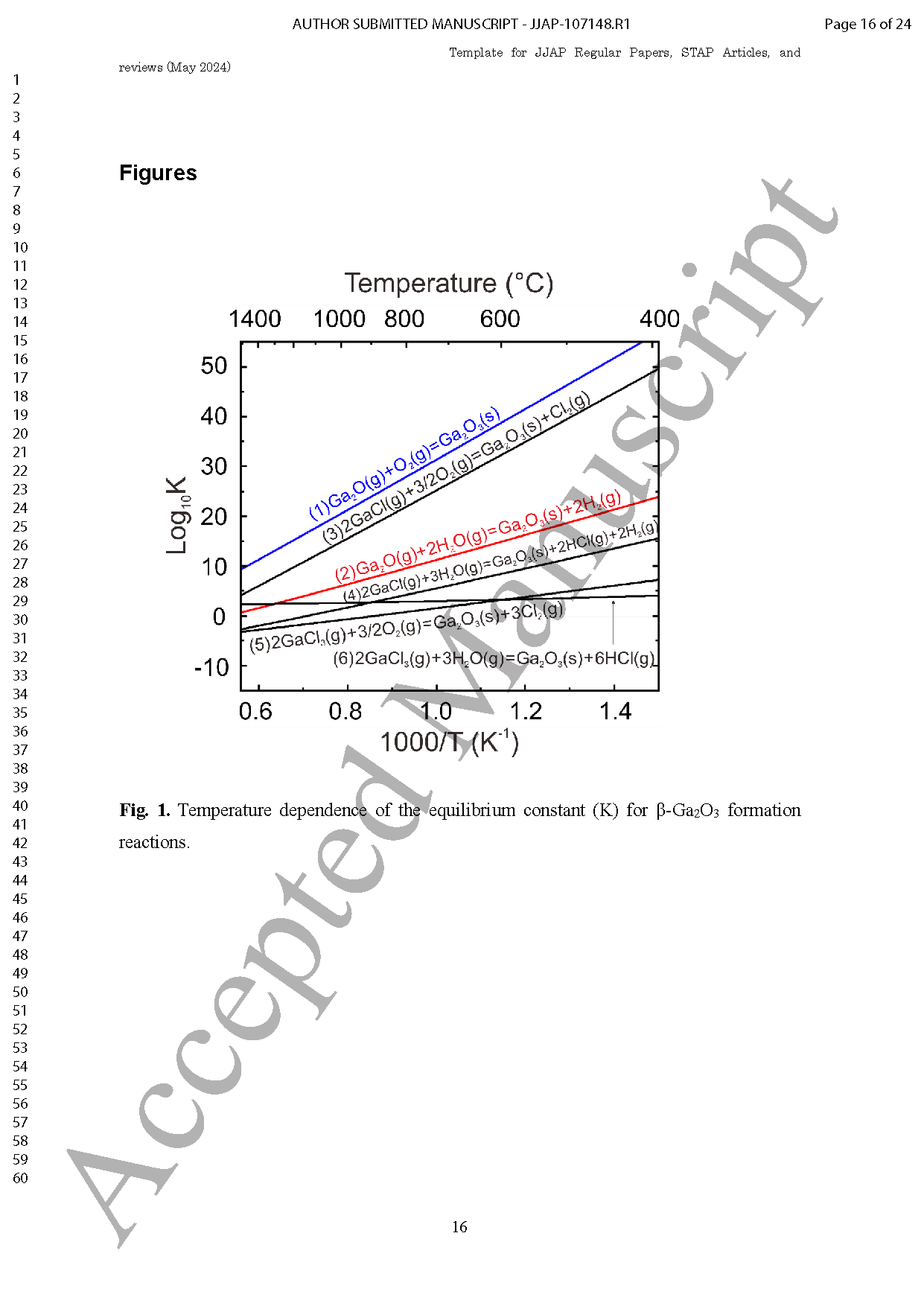
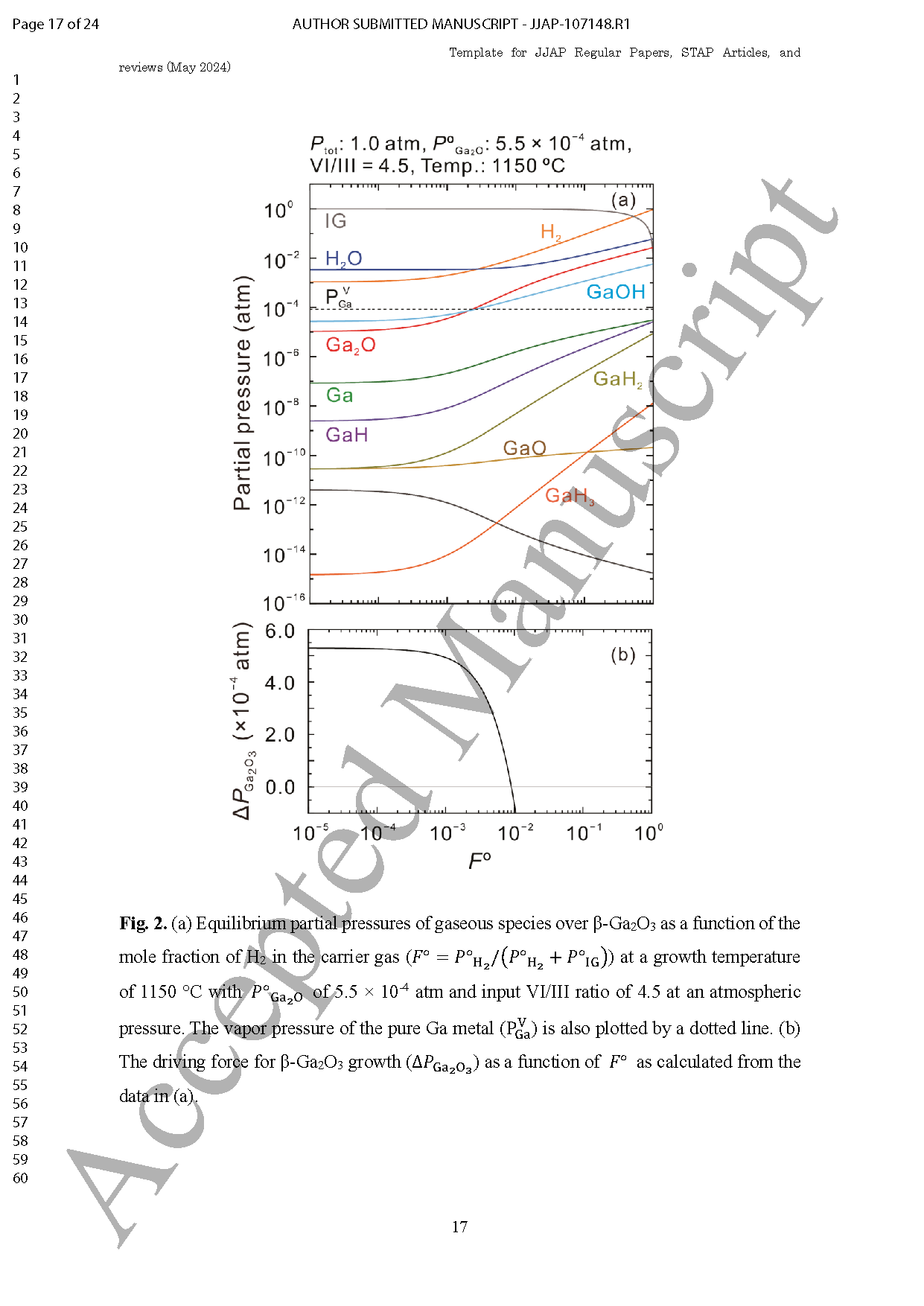
Fig. 1. (a) Equilibrium partial pressures of gaseous species over β-Ga2O3 as a function of the mole fraction of H2 in the carrier gas (FO=POH2+POIG) at a growth temperature of 1150 °C with POGa2O of 5.5 ×10-4 atm and input VI/III ratio of 4.5 at an atmospheric pressure. The vapor pressure of the pure Ga metal (PVGa) is also plotted by a dotted line. (b) The driving force for β-Ga2O3 growth (△PGa2O3) as a function of FO as calculated from the data in (a).
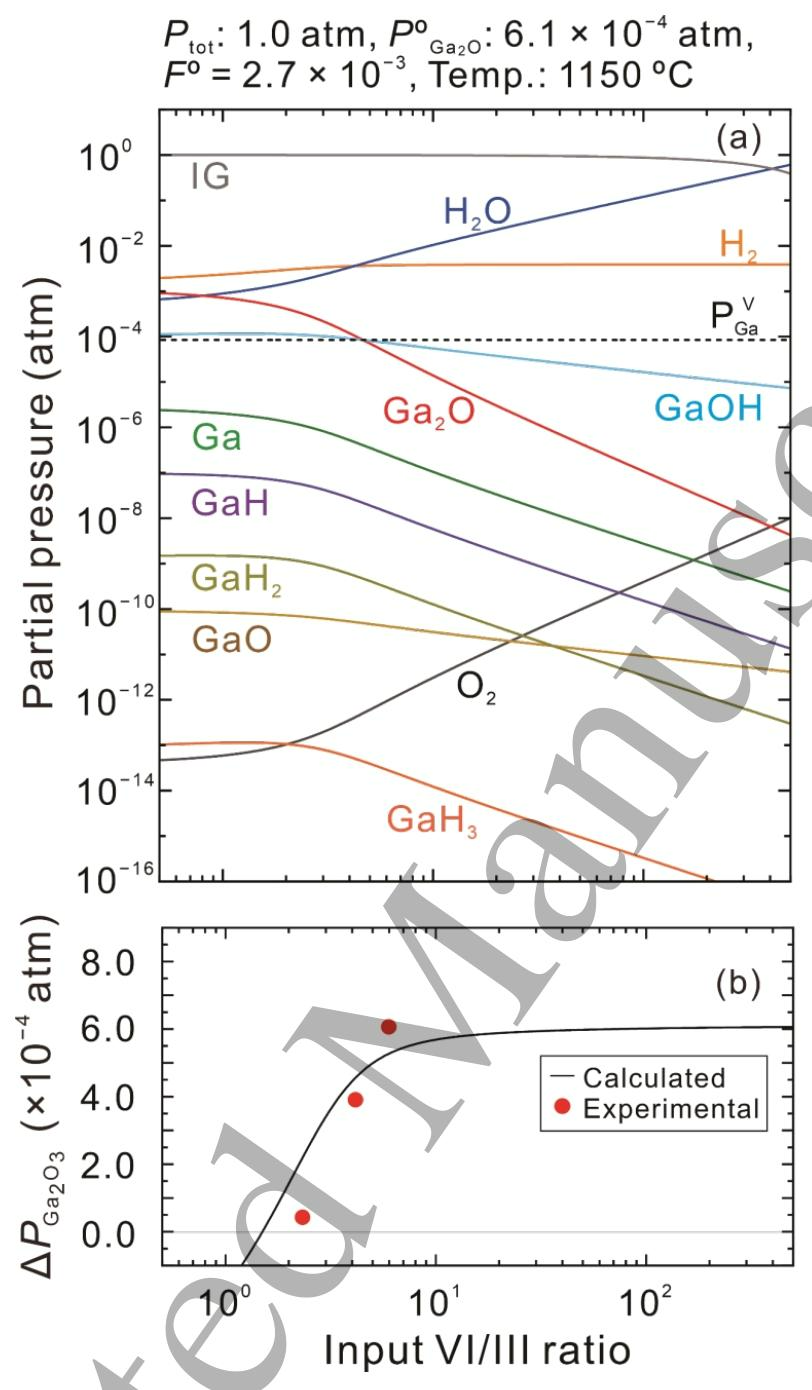
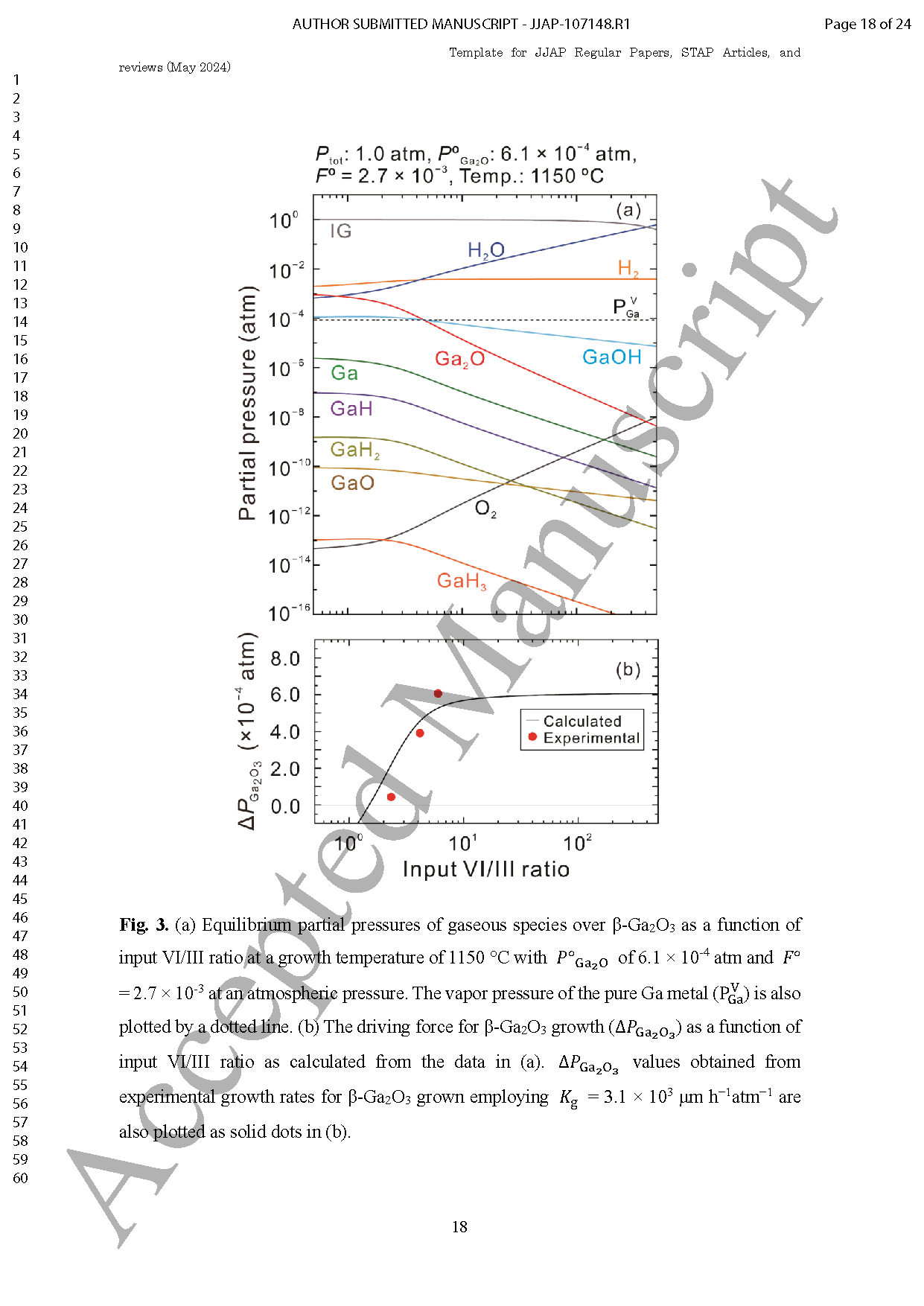
Fig. 2 (a) Equilibrium partial pressures of gaseous species over β-Ga2O3 as a function of input VI/III ratio at a growth temperature of 1150 °C with POGa2O of 6.1 ×10-4 atm and FO=2.7 ×10-3 at an atmospheric pressure. The vapor pressure of the pure Ga metal PVGa also plotted by a dotted line. (b) The driving force for β-Ga2O3 growth △PGa2O3 as a function of input VI/III ratio as calculated from the data in (a). △PGa2O3 values obtained from experimental growth rates for β-Ga2O3 grown employing Kg=3.1 ×103 μm h-1 atm-1 are also plotted as solid dots in (b).
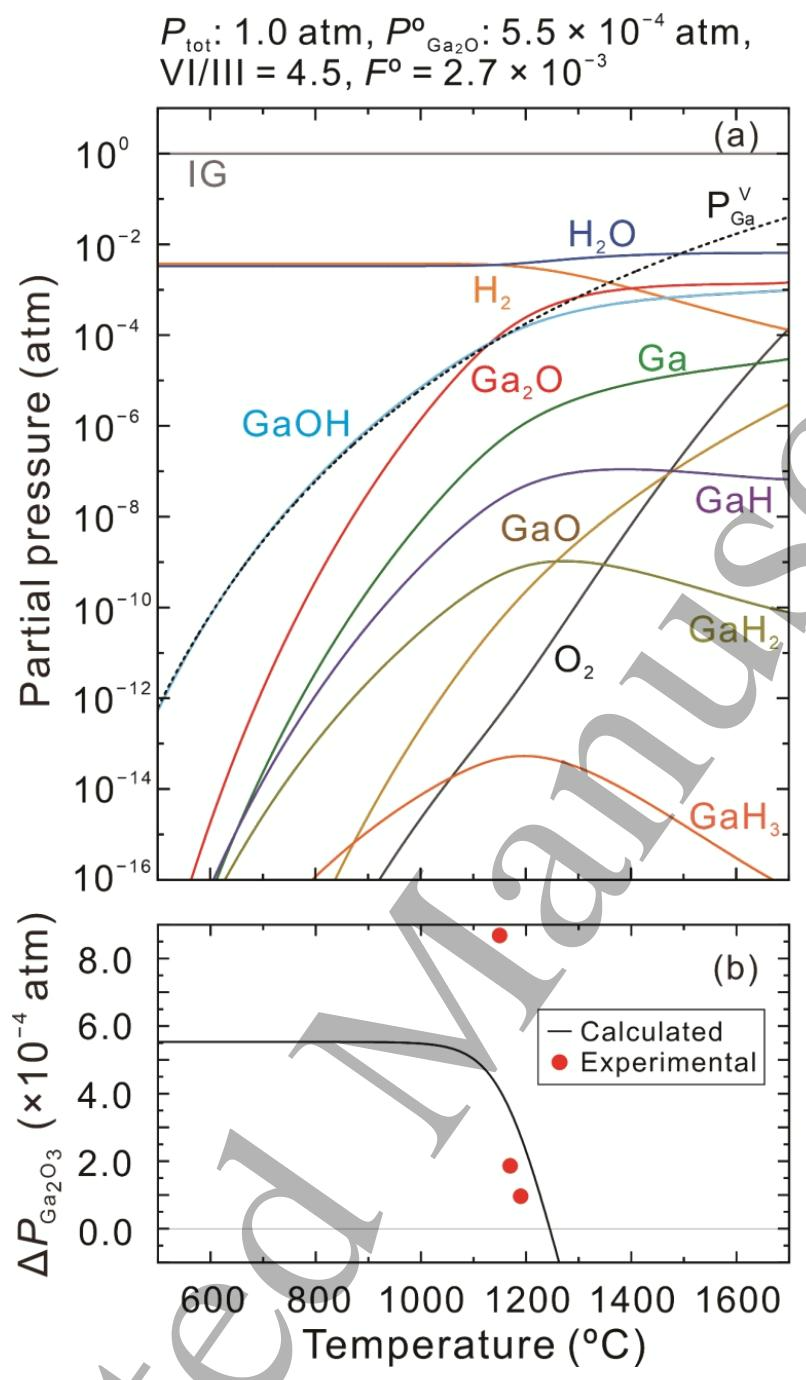
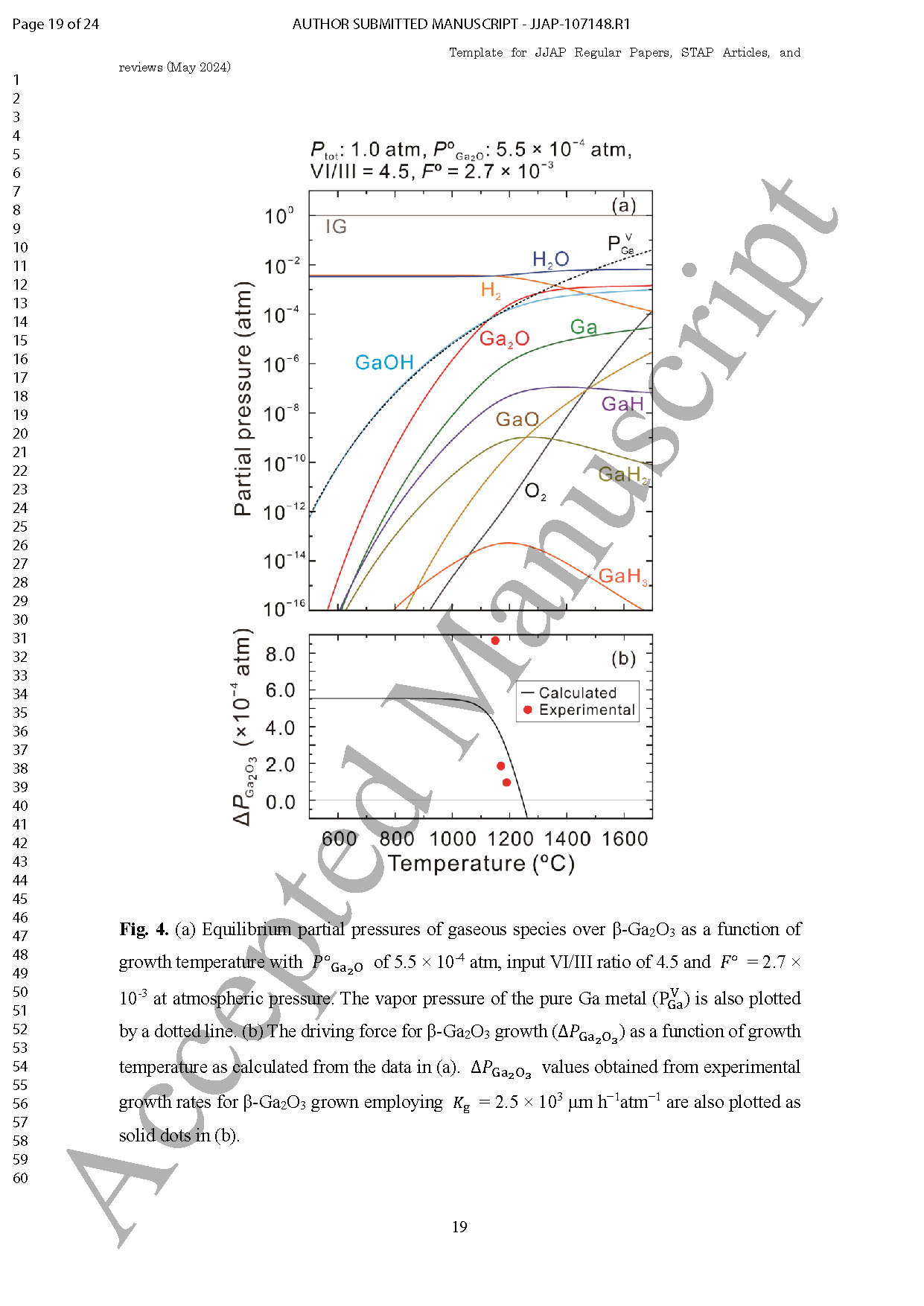
Fig. 3. (a) Equilibrium partial pressures of gaseous species over β-Ga2O3 as a function of growth temperature with POGa2O of 5.5×10-4 atm, input VI/III ratio of 4.5 and FO =2.7×10-3 at atmospheric pressure. The vapor pressure of the pure Ga metal PVGa is also plotted by a dotted line. (b) The driving force for β-Ga2O3 growth △PGa2O3 as a function of growth temperature as calculated from the data in (a). △PGa2O3 values obtained from experimental growth rates for β-Ga2O3 grown employing Kg=2.5×103 μm h-1 atm-1 are also plotted as solid dots in (b).
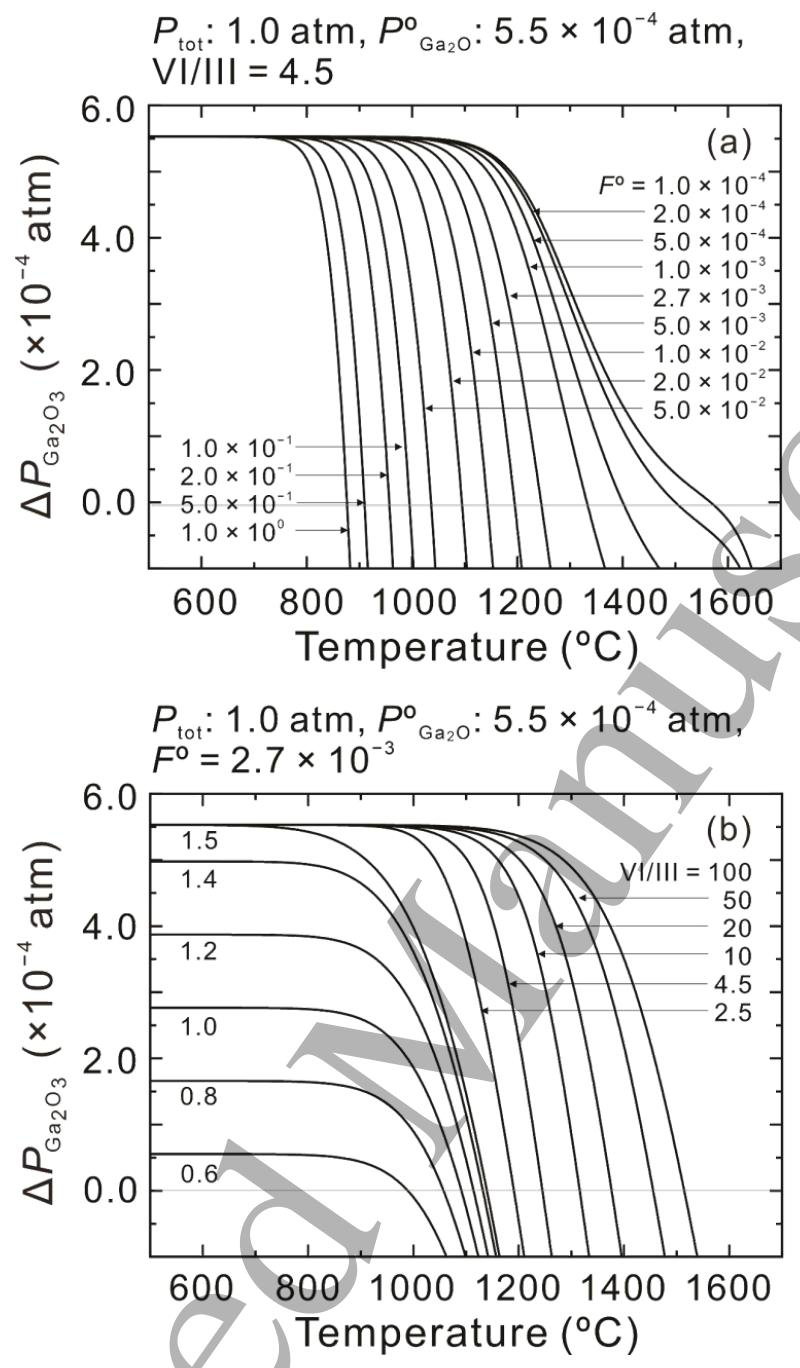
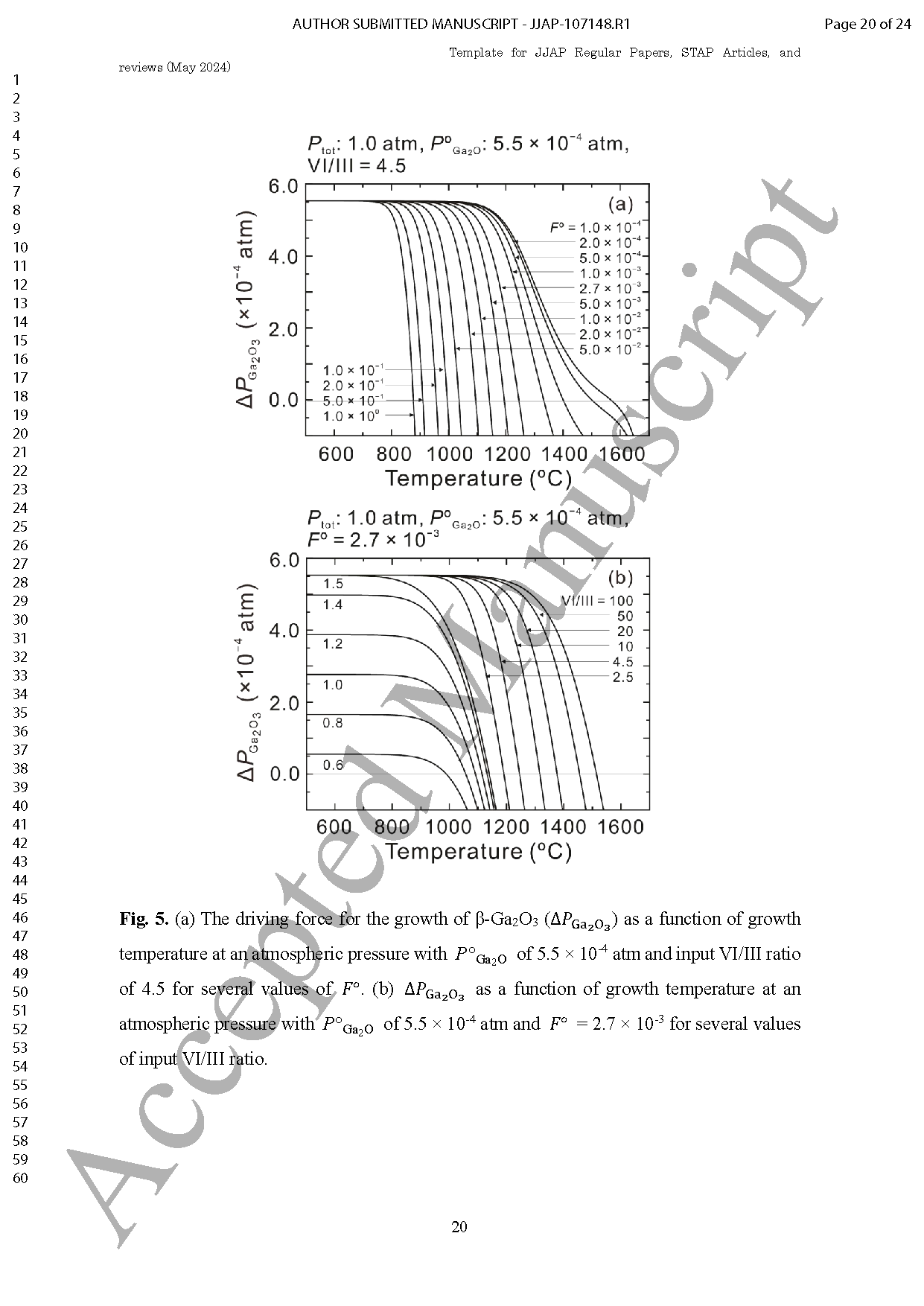
Fig. 4. (a) The driving force for the growth of β-Ga2O3 (△PGa2O3) as a function of growth temperature at an atmospheric pressure with POGa2O of 5.5×10-4 atm and input VI/III ratio of 4.5 for several values of FO .(b) △PGa2O3 as a function of growth temperature at an atmospheric pressure with POGa2O of 5.5×10-4 atm and FO =2.7 ×10-3 for several values of input VI/III ratio.
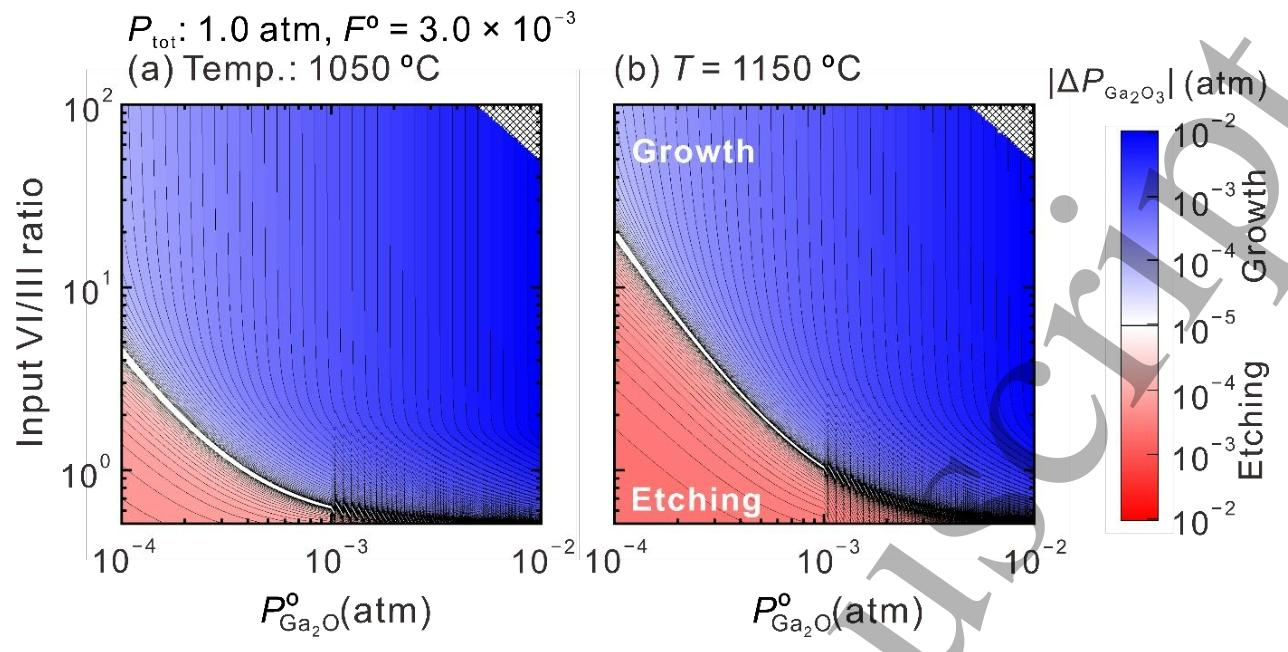
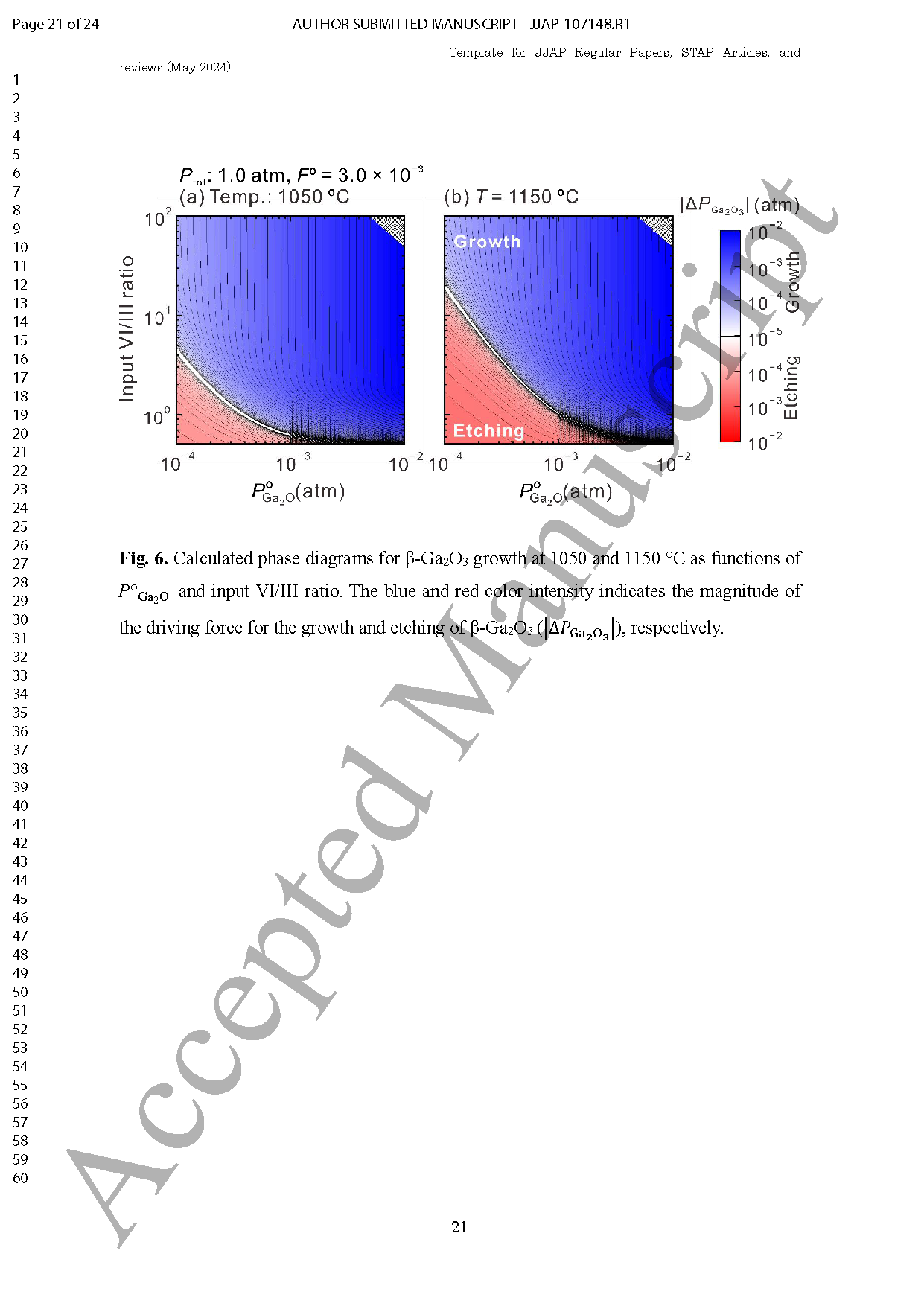
Fig. 5. Calculated phase diagrams for β-Ga2O3 growth at 1050 and 1150 °C as functions of POGa2O and input VI/III ratio. The blue and red color intensity indicates the magnitude of the driving force for the growth and etching of β-Ga2O3 (|△PGa2O3|), respectively.
DOI:
doi.orq/10.35848/1347-4065/ae54ed