【Others Papers】Hydroxyl-facilitated efficient propane dehydrogenation over bare Ga₂O₃ via altering reaction pathway
日期:2026-03-10阅读:171
Researchers from the Beijing Institute of Technology have published a dissertation titled "Hydroxyl-facilitated efficient propane dehydrogenation over bare Ga2O3 via altering reaction pathway" in Journal of Catalysis.
Background
The efficient utilization and conversion of light alkanes is significantly important for realizing a green and low-carbon society. As an important industrial and model reaction, non-oxidative propane dehydrogenation (PDH), acting as an alternative and on-purpose propene production technology to traditional oil-based technologies (steam or catalytic cracking of different oil fractions), has received increasing attention due to the advantages of abundant light alkane resource, higher propene selectivity, and easy separation of reaction products. The growing demand for propene has also driven the rapid development of the PDH process. In the aspect of fundamental research, PDH has been recognized as a platform reaction for studying the C–H bond activation over various types of catalysts including noble metal, and metal oxide-based materials.
Abstract
Designing suitable catalysts for catalytic processes involving alkanes and efficiently activating C−H bond in light alkanes are both theoretically and practically significant. The present study shows that bare Ga2O3 without any supported species or dopants can effectively catalyze the PDH reaction. A clear structure–activity relationship based on crystallite size, acid density, and hydroxyl group density is established, the smaller the crystallite size, the higher the activity. Ga2O3 with a smaller crystallite size owns a higher concentration of hydroxyl species and oxygen vacancies. Comprehensive experimental investigations and DFT calculations indicate that the presence of oxygen vacancy in Ga2O3 decreases the apparent activation energy of PDH compared with the pristine Ga2O3 surface. Importantly, the existence of hydroxyl groups on the Ga2O3 surface can significantly improve the ability to activate C−H bond by altering the PDH reaction from a non-oxidative to the oxidative pathway at the initial stage. It is expected that these findings offer fundamental insights into regulating the physicochemical properties of metal oxides for efficient C−H bond activation and hydrogenation reactions.
Highlights
● A series of bare Ga2O3 with different crystallite size were synthesized by different methods.
● The smaller the crystallite size of Ga2O3, the higher the PDH activity.
● The structure–activity relationship and reaction mechanism were investigated by experimental and theoretical studies.
● The presence of hydroxyl groups on the Ga2O3 surface can significantly improve the ability to activate C−H bond.
Conclusion
In summary, this study investigated the structure–activity relationship of bare Ga2O3 for PDH reaction by combining complementary characterization techniques with DFT calculations. It is found that the crystallite size has strong effects on the microstructure, acidic, and surface properties of Ga2O3. The smaller the size of crystallite, the higher the concentration of acid sites and hydroxyl species, and therefore the higher the rate of propene formation in PDH. The presence of hydroxyl species on the Ga2O3 surface alters the reaction pathway of PDH from a non-oxidative pathway to the oxidative pathway at the initial stage concerning the first C− H bond activation, significantly decreasing the reaction barrier of C− H bond activation. In addition, Ga2O3 with smaller crystallite size owns a high concentration of oxygen vacancy forming Ga2O3–Ov sites, which shows a lower apparent activation barrier than that of Ga2O3 and Ga2O3–H surfaces. Moreover, Ga2O3 with different crystallite sizes exhibits very high propene selectivity (>97%) and the selectivity of cracking products is low over a wide range of propane conversions. It is believed that the present study contributes to deeply understanding the C−H bond in light alkanes and promoting the development of Ga2O3-based materials in PDH reaction.
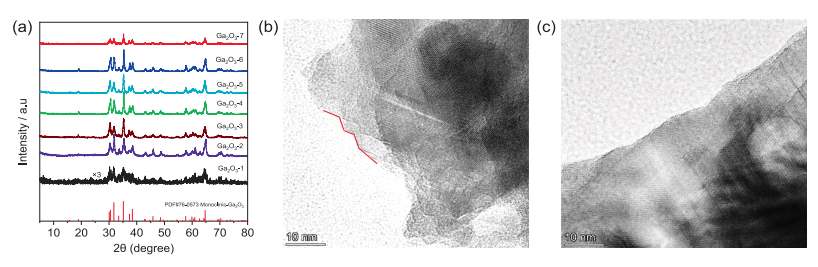
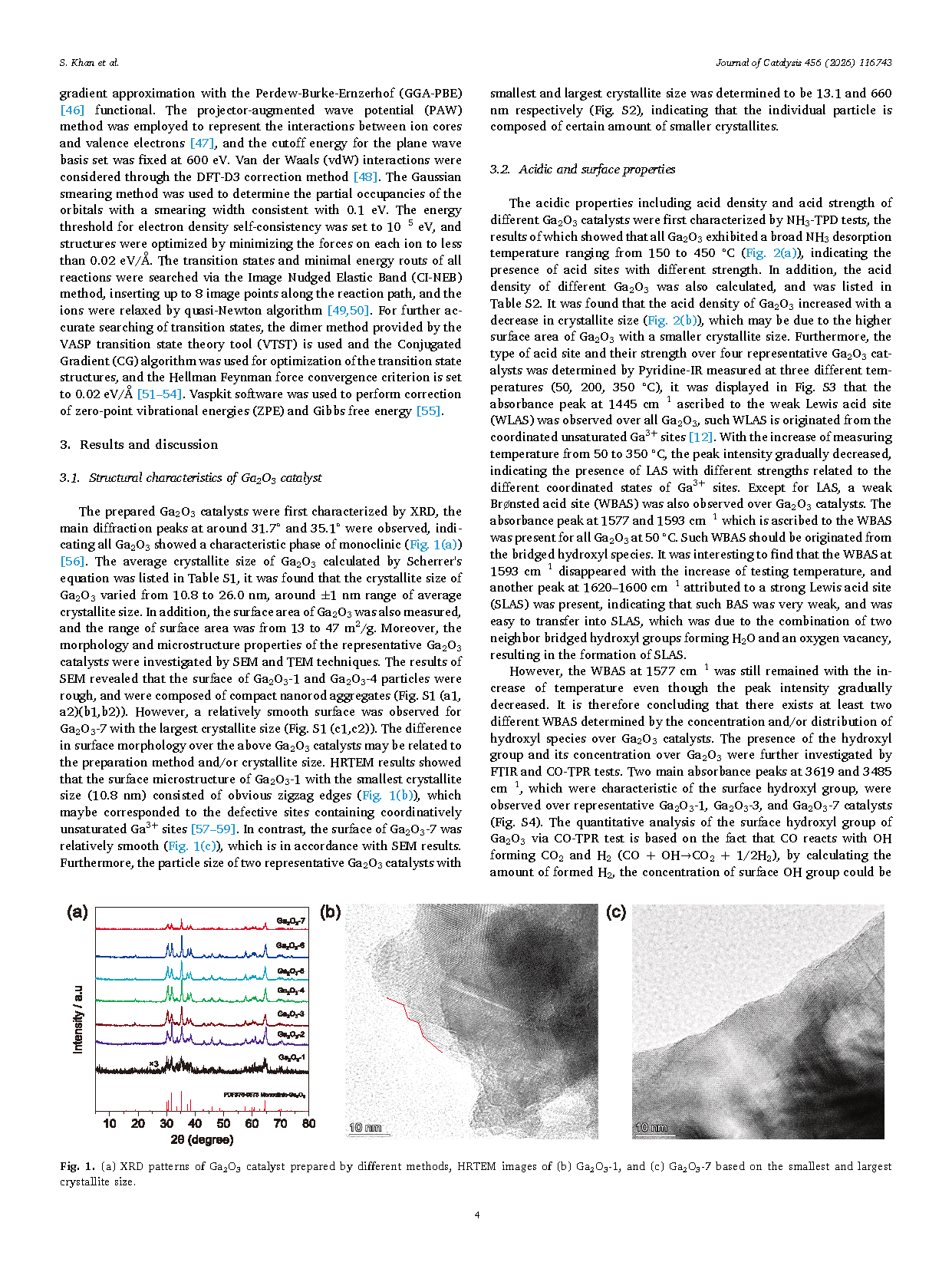
Fig. 1. (a) XRD patterns of Ga2O3 catalyst prepared by different methods, HRTEM images of (b) Ga2O3-1, and (c) Ga2O3-7 based on the smallest and largest crystallite size.
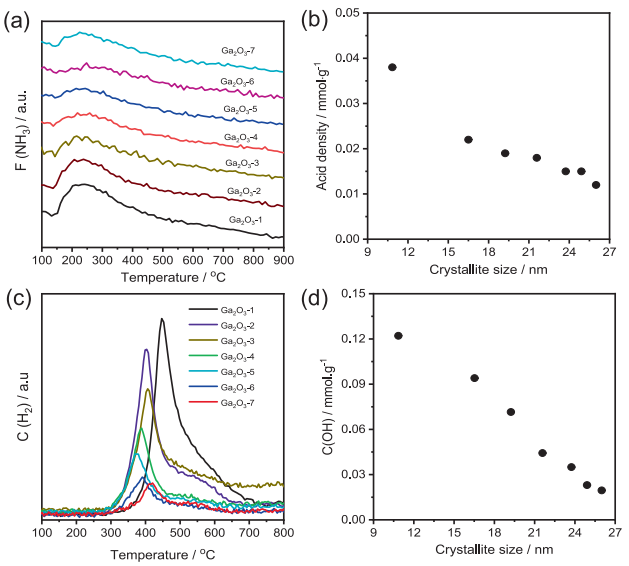
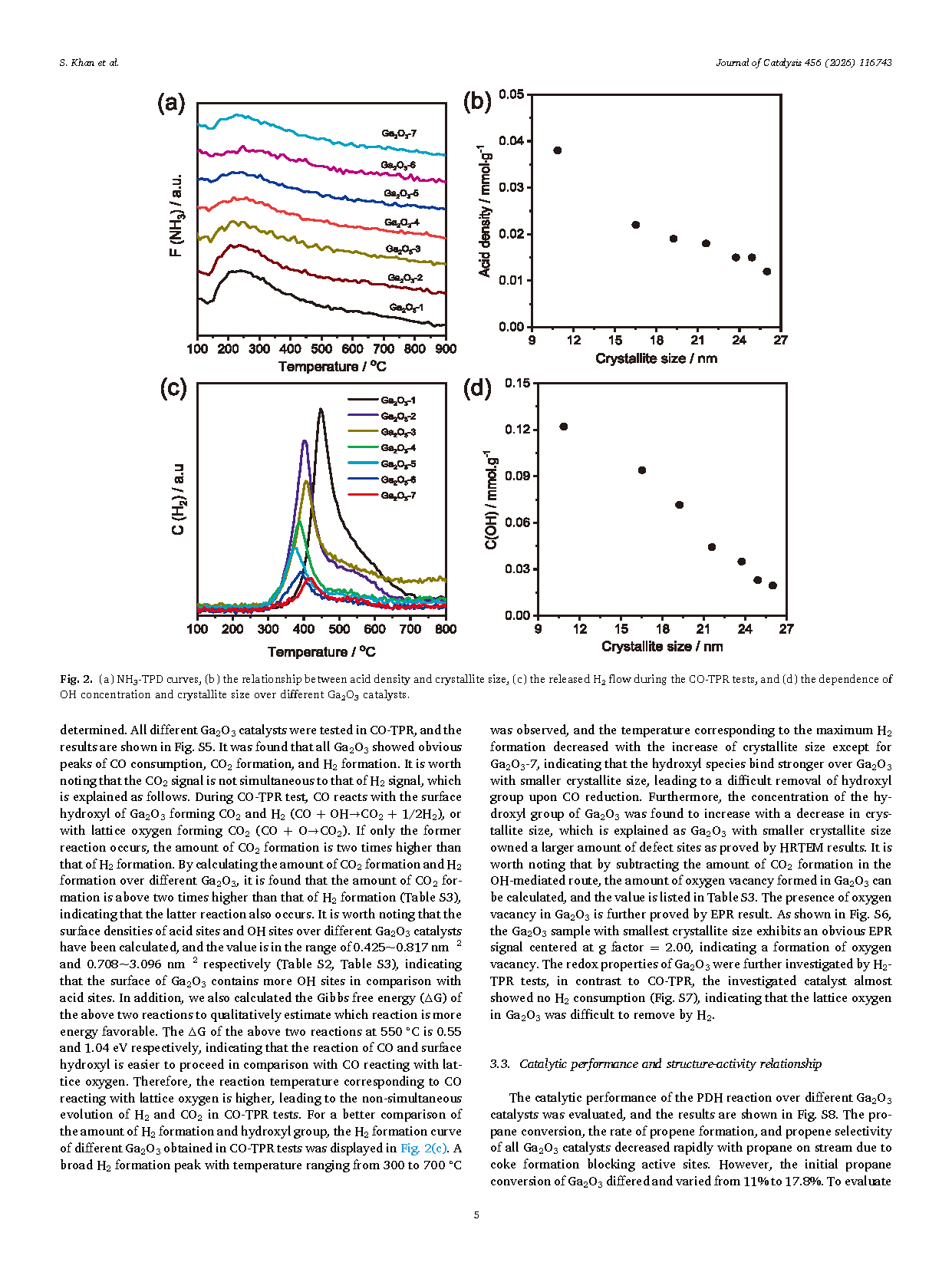
Fig. 2. (a) NH3-TPD curves, (b) the relationship between acid density and crystallite size, (c) the released H2 flow during the CO-TPR tests, and (d) the dependence of OH concentration and crystallite size over different Ga2O3 catalysts.
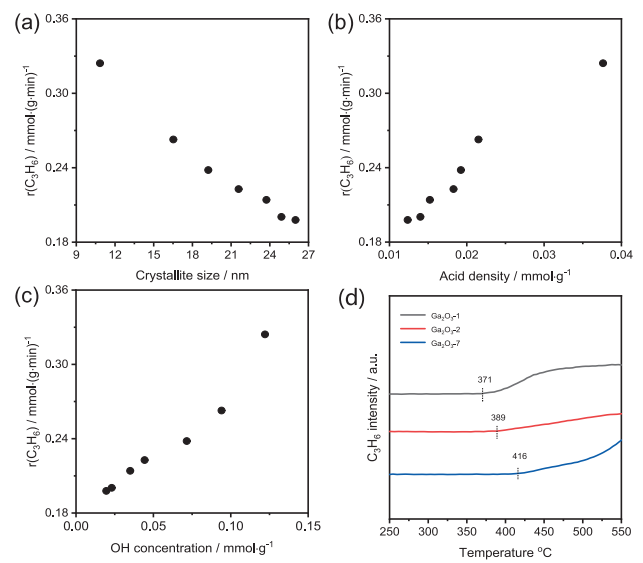
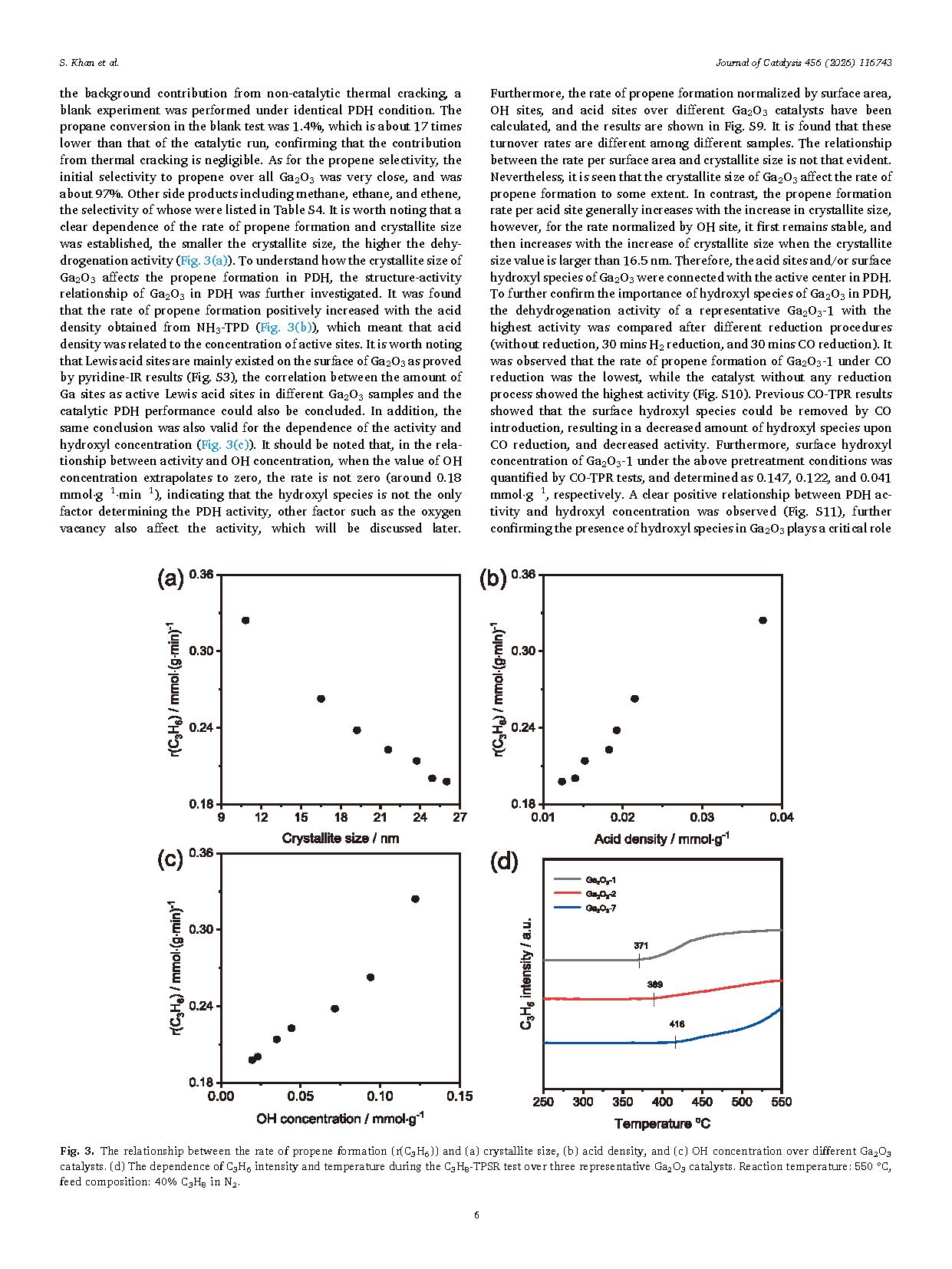
Fig. 3. The relationship between the rate of propene formation (r(C3H6)) and (a) crystallite size, (b) acid density, and (c) OH concentration over different Ga2O3 catalysts. (d) The dependence of C3H6 intensity and temperature during the C3H8-TPSR test over three representative Ga2O3 catalysts. Reaction temperature: 550 °C, feed composition: 40% C3H8 in N2.
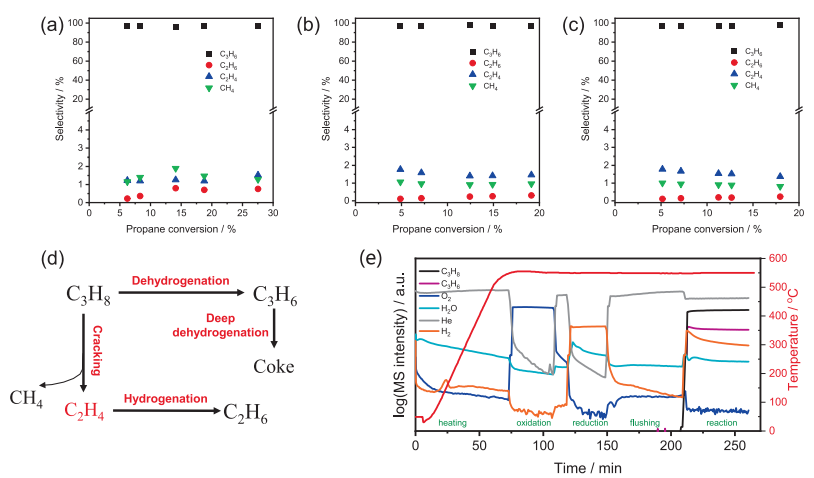
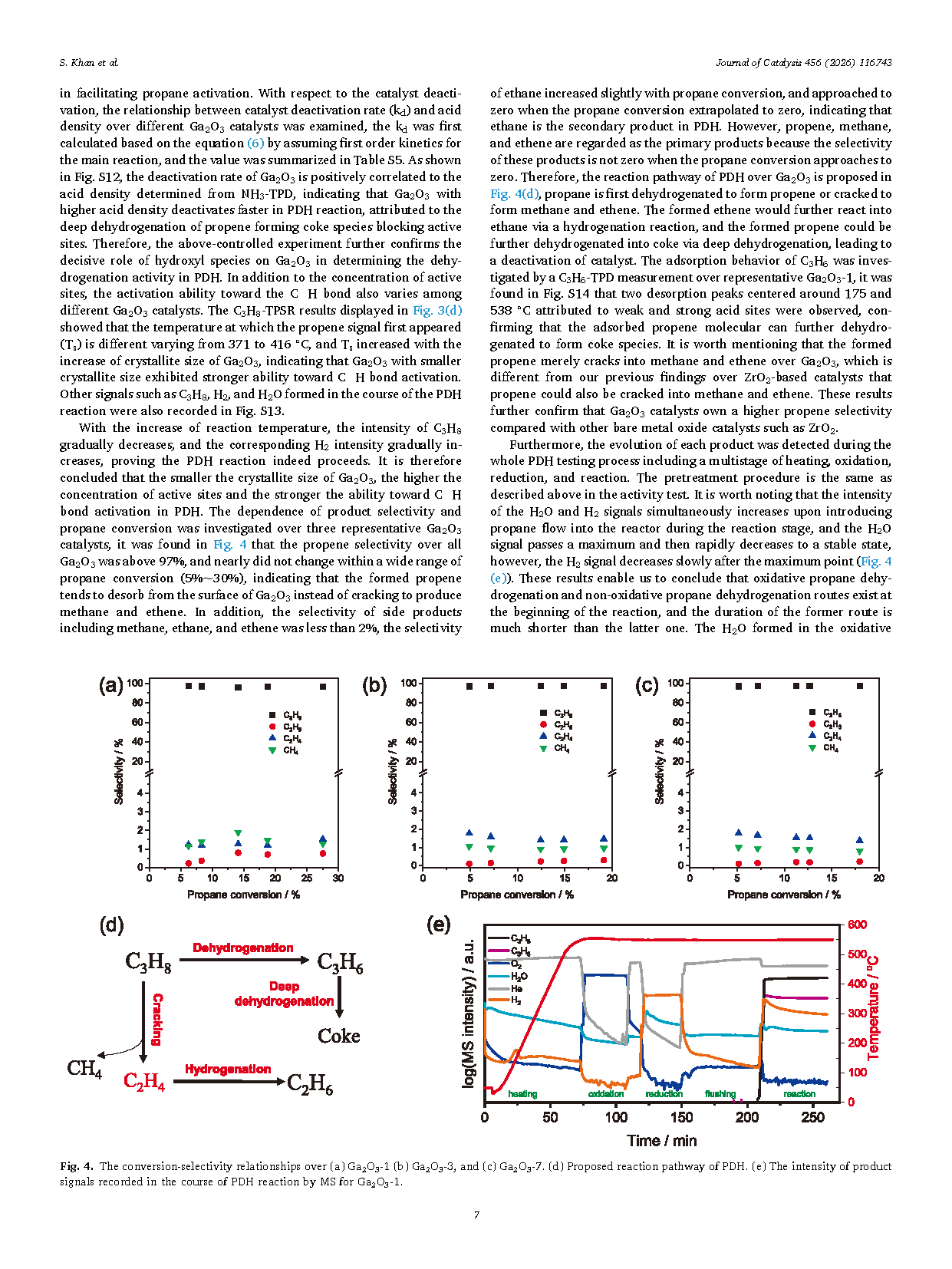
Fig. 4. The conversion-selectivity relationships over (a) Ga2O3-1 (b) Ga2O3-3, and (c) Ga2O3-7. (d) Proposed reaction pathway of PDH. (e) The intensity of product signals recorded in the course of PDH reaction by MS for Ga2O3-1.
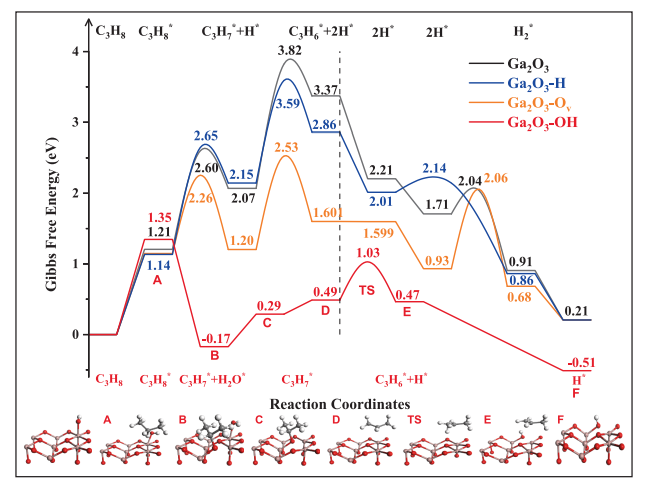
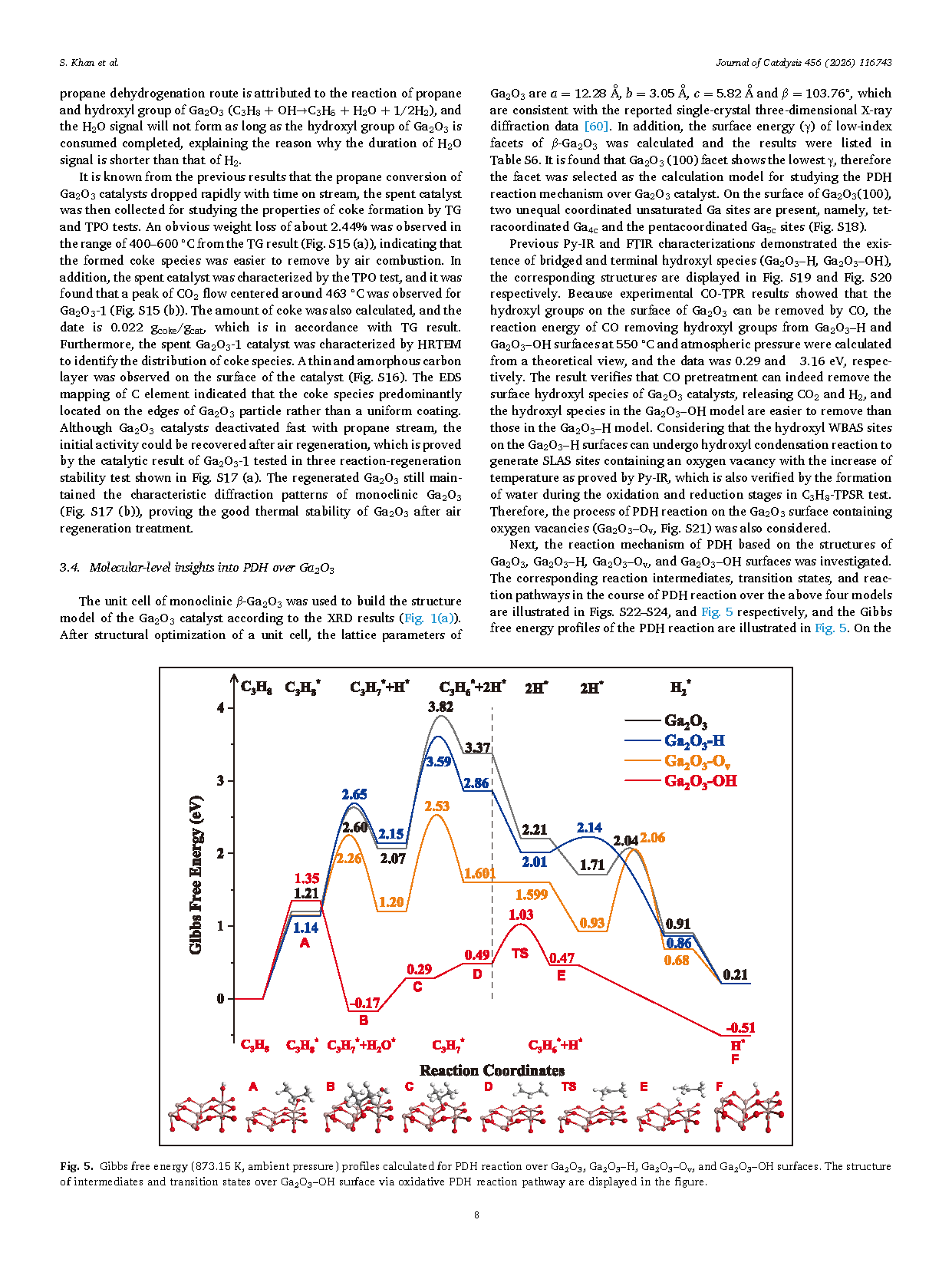
Fig. 5. Gibbs free energy (873.15 K, ambient pressure) profiles calculated for PDH reaction over Ga2O3, Ga2O3–H, Ga2O3–Ov, and Ga2O3–OH surfaces. The structure of intermediates and transition states over Ga2O3–OH surface via oxidative PDH reaction pathway are displayed in the figure.
DOI:
doi.org/10.1016/j.jcat.2026.116743