

【Knowledge Discover】Basic Principles of X-ray Photoelectron Spectroscopy (XPS) and Its Application in Gallium Oxide Material Analysis
日期:2026-05-08阅读:41
X-ray Photoelectron Spectroscopy (XPS), also known as Electron Spectroscopy for Chemical Analysis (ESCA), is one of the core techniques in the field of material surface characterization. Based on the photoelectric effect, XPS enables reliable analysis of the elemental composition and chemical states of elements on a material’s surface.
The fundamental principle of XPS originates from Einstein’s photoelectric effect. When X-rays with fixed energy irradiate the sample surface, inner-shell electrons are excited and emitted from atoms. By measuring the kinetic energy of the emitted photoelectrons, the binding energy of the inner-shell electrons can be determined. The corresponding fundamental equation is:
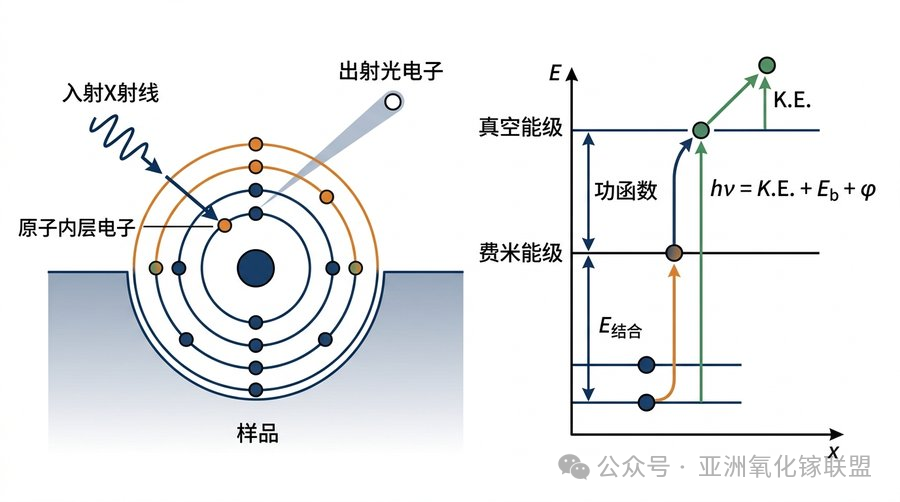
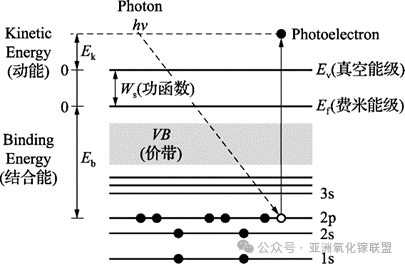
When the excitation source energy is fixed, the kinetic energy of the emitted photoelectrons depends only on the elemental species and the atomic orbital from which the electrons are excited. Therefore, the elemental composition of a material can be qualitatively identified based on the binding energies of the photoelectrons. By measuring the binding energies of different elements in a sample, the surface chemical composition, chemical states, and relative contents of elements can be determined, enabling both qualitative and quantitative analysis.
XPS probes inner-shell electrons, which do not directly participate in chemical bonding. However, these electrons are influenced by the distribution of valence electrons outside the nucleus. Changes in the chemical environment alter the valence electron density, which in turn changes the shielding effect experienced by inner-shell electrons, ultimately resulting in binding energy shifts. This is the fundamental reason why XPS can characterize chemical bonding states.
XPS spectra are generally divided into survey spectra and high-resolution spectra. Survey spectra are mainly used to identify the elemental composition and approximate content of elements in a sample (except H and He). However, the signals obtained from survey scans are relatively coarse and mainly provide rough elemental information. For elements with low concentrations, the signal-to-noise ratio is often poor. As a result, survey spectra usually provide only surface composition information and cannot accurately reveal chemical states or molecular structure information.
In addition to survey scans, XPS measurements often include detailed analysis of specific elements, producing high-resolution spectra. By analyzing the peak positions in high-resolution spectra, the chemical states of elements can be identified. Furthermore, comparing chemical shifts before and after sample treatment can reveal changes in surface chemical states or electronic interactions between surface elements. In general, when an element loses electrons, its binding energy shifts toward higher binding energy.
Currently, in the field of Gallium Oxide materials, XPS is commonly employed to investigate material properties, particularly for studying oxygen vacancies and related surface chemical characteristics.
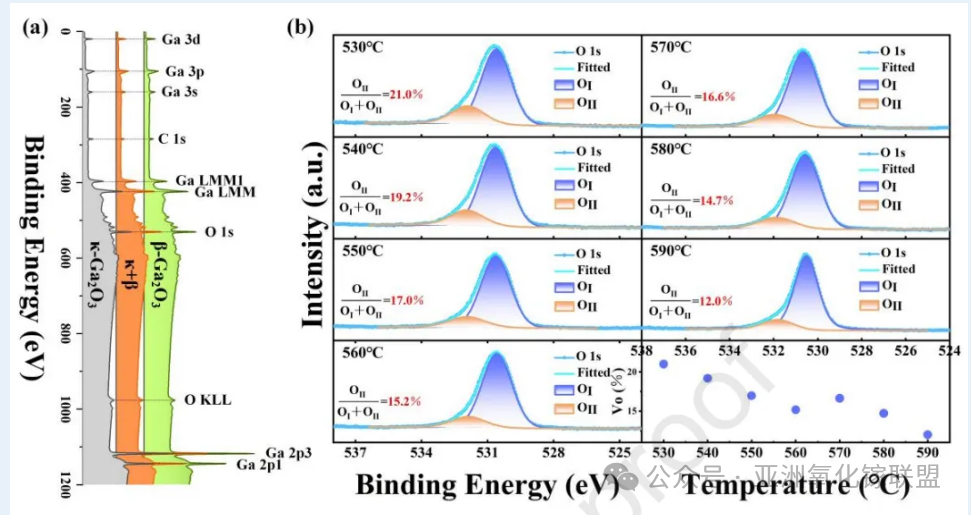
Zhang J, Song Y, Cui W, et al. Geometric and Thermal Control of κ-to β-Ga2O3 Phase Engineering for High-Performance Solar-Blind Photodetector[J]. Materials Today Physics, 2026: 102105.
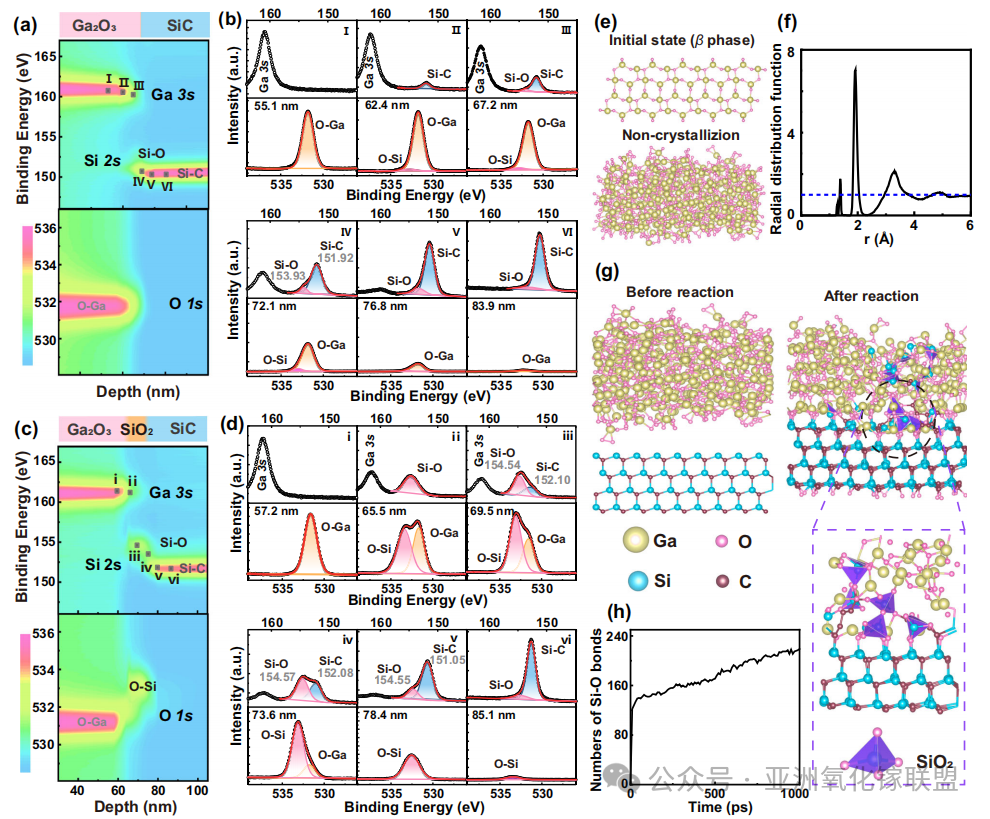
Yi Shen,Xin Qi,Yuan Li,Yunduo Guo,Qimin Huang,Wen Dai,Qilong Yuan,Lin Gu,Chengxi Ding,Wen-Jie Chen,Mingyang Yang,Zhenglin Jia,Cheng-Te Lin,Nan Jiang,Fangyuan Sun,Qingchun Jon Zhang,Hong-Ping Ma,Improving interfacial thermal conductivity by constructing covalent bond between Ga2O3 and SiC,Nature Communications,(2025) 16:10723


